Hero

Your future starts at M State
Plan your visit today to one or more of our four campuses in Detroit Lakes, Fergus Falls, Moorhead and Wadena
Paragraphs
Why M State?
98%
placement rate
#1
community college in MN
70+
programs and degrees
4
campuses, plus online
24
average class size
M State programs are in fields employers are hiring for.

Accounting
You'll get all the accounting skills and knowledge you need to begin your career with confidence with this degree program.

Accounting Clerk
You’ll be on your way to a career in financial accounting in just two semesters with this diploma program.

Accounting Transfer Pathway
Enjoy all the advantages of an affordable smaller college as you lay the educational foundation for advanced studies in accounting.

Advanced HVAC/R
This diploma program gives you the next-level training you need to become qualified for specialized work in the commercial refrigeration industry.
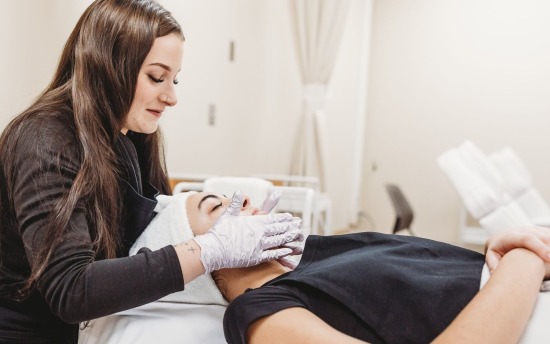
Advanced Practice Esthiology
Licensed estheticians and cosmetologists: expand your skills and licensure to offer advanced skincare services and procedures.

Agribusiness
Cultivate a fruitful career in agricultural sales, management or technical work with this degree program.

American Sign Language
Strengthen your sign language skills and gain a better understanding of Deaf culture, history and experiences with our degree and certificate options.

Architectural Drafting and Design
You’ll be ready to begin a career with an architectural or engineering firm or construction industry-related contracting, manufacturing or distribution company after completing M State's Architectural Drafting and Design program.

Art Transfer Pathway
Be as creative about your college plan as you are about your artwork: Start your studies at M State and then seamlessly transfer to a university to finish your four-year degree.
Associate Degree Nursing
Meet the requirements for professional licensure as a registered nurse with this hands-on degree program that features multiple clinical experiences.
Associate of Arts - Liberal Arts and Sciences Transfer Pathway
This popular, flexible transfer program gives you the freedom to concentrate in the liberal arts field of your choice.

Associate of Arts - Social Science Emphasis
Prepare for a career or further study in sociology, social work and human services with this degree program.

Automotive Service Technology
Work on cars and light trucks side-by-side with expert instructors at our on-site lab to diagnose, repair and prevent mechanical issues. Both a degree and diploma option are available.
Biology Transfer Pathway
This degree program will give you the foundational coursework you need to seamlessly transfer to a university to pursue an advanced degree in biology.

Business
Concentrate in the business field of your choice to prepare for a career in retail, insurance, sales and other in-demand professions with this degree program.

Business Administration
This two-term diploma program can stand on its own or be seamlessly integrated into our Business degree program.

Business Transfer Pathway
Start your studies in business at M State and then seamlessly transfer to a university to complete your bachelor’s degree.

Child and Adolescent Therapeutic Behavioral Health
Become certified to begin a rewarding career in supervised behavioral health care for children and adolescents.
Computer Programming
Prepare for a position in a lucrative field with this nationally-recognized degree program. You'll learn to design and develop computer software and information systems.

Construction Management
Build the construction and leadership skills most wanted by industry employers with this across-the-board degree program.

Cosmetology
Hands-on practice at our campus salon helps you build skills in hair, skin and nail care – and earn clinic hours to put toward your licensure.

Criminal Justice
Graduates of this degree program are in high demand thanks to its regional reputation for high quality, along with growing career opportunities in law enforcement.

Cybersecurity
Learn how to keep networks secure and data safe – skills today's employers can't get enough of – with our Cybersecurity degree and certificate programs.
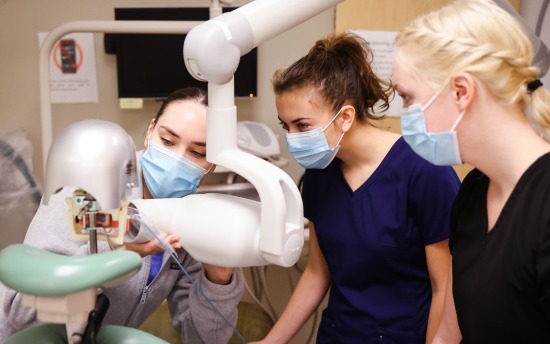
Dental Assisting
Gain the knowledge and hands-on skills experience to work closely and confidently with dentists during direct patient care, including diagnostic, preventative and operative procedures.
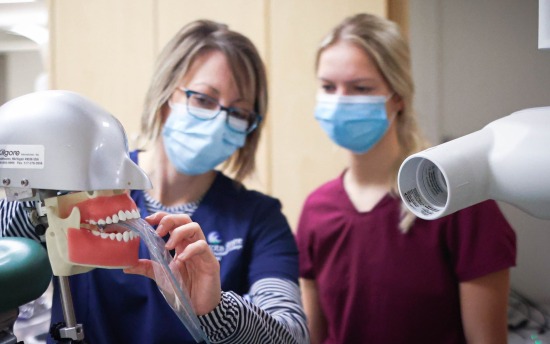
Dental Hygiene
Develop the skills to enjoy a rewarding career in comprehensive dental hygiene care with this hands-on degree program.

Design and Engineering Technology
Enter the workforce or pursue advanced studies after completing this associate degree program. A certificate option is also available.

Diesel Equipment Technology
Tools supplied to second-year students and four different degree and diploma tracks, including a sponsored degree, put this program a cut above.

Digital Marketing
Master the latest digital tools and technology to create engaging, inspiring marketing campaigns with this degree program.

Early Childhood
Open your own in-home childcare business or work at a childcare center upon completion of this certificate program.

Early Childhood Education Transfer Pathway
Start your education at M State and then seamlessly transfer to a university to pursue an advanced degree in Early Childhood Education.

Electrical Line Worker Technology
This popular program has a reputation for excellence. You'll get all the hands-on skills practice you need at our 90-acre training facility. Both a degree and diploma option are available.

Electrical Technology
Job placement rates are typically 98-100% for this high-demand diploma program, which couples electrical theory coursework with hands-on practice.

Elementary Education Transfer Pathway
Upon completion of this 60-credit M State degree, you'll be a thoroughly prepared elementary teacher candidate ready to create engaging and inclusive classrooms.

Engineering
Enjoy all the advantages of an affordable smaller college as you lay the educational foundation for advanced studies in engineering with this degree program.
Entrepreneurial Certificate
The practical, professional knowledge you need to start and run your own successful business.

Environmental Science
This degree program will prepare you for a rewarding career in the natural sciences. You may also choose to pursue further studies at a four-year college or university.

Esthetician
The knowledge, exam prep and hands-on skills practice you need to earn your licensure and begin a rewarding career in skin care.

Global and Intercultural Studies
Liberal arts degree-seeking students can gain a global perspective and meet their elective requirements with this embedded certificate program.
Graphic Design
Earn your certificate or diploma to grow your passion for design and enjoy an exciting, creative career.

Health Information Technology/Coding
Care for patients by caring for their medical data with this comprehensive, reputable degree program.

Health Sciences Broad Field
Start your studies at M State and then seamlessly transfer to a university to pursue an advanced degree in health sciences.
History Transfer Pathway
This degree provides the foundational knowledge you need to make a smooth and successful transition into a bachelor's degree program in history.

Human Resources
Degree and certificate programs will help you develop your business acumen and human relations skills to become a trusted HR professional.

HVAC/R
Qualified HVAC/R workers are in high demand, and our hands-on diploma program will fully prepare you for a successful career in the industry.
Information Technology
The technology training you need to begin a career in one of the fastest-growing job fields in the nation. Choose between two different degree options.

Integrated & Professional Studies
A degree program uniquely designed and developed by you, for you – with college credits given for relevant prior work experience.

Limited Scope Radiography
You’ll be ready to perform a variety of radiography exams on patients upon completion of this in-demand diploma program.

LPN to Associate Degree Nursing Bridge
This degree program is designed for licensed practical and vocational nurses who want to become registered nurses.
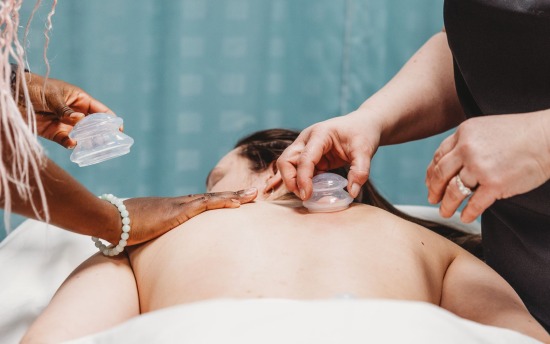
Massage Therapy
Take on a truly hands-on career: complete this flexible diploma program and support people’s health with therapeutic massage.

Medical Coding
Fill an essential in-demand administrative role in healthcare with this specialty diploma program.

Medical Laboratory Technician
Test biological specimens at healthcare center laboratories with this associate of applied science degree that also prepares you to take the MLT Board of Certification exam.
Music
Choose a vocal or instrumental focus and gain live performance experience with this Associate of Fine Arts degree.

Nail Technician
The knowledge and hands-on skills practice you need to really nail a career as a manicurist and pedicurist.

Nursing Assistant
Become a CNA (certified nursing assistant), fast, by completing this 77-hour course through M State's Workforce Development Solutions.

Payroll Specialist
Professionally prepare and report payroll, a specialty within the field of accounting, with this two-term diploma program.
Phlebotomy Technician
Enter this in-demand healthcare field fast with our one-semester certificate program, which includes a valuable five-week clinical experience.

Plumbing Technology
Benefit from this diploma program's hands-on plumbing experiences, an internship in the field, and potential industry incentives.

Political Science Transfer Pathway
Start your studies in political science at M State and then seamlessly transfer to a university to complete an advanced degree.

PowerSports Technology
Degree, diploma and certificate options all provide industry-recognized training in the service and repair of small engines for motorcycles, snowmobiles, ATVs and watercraft.

Practical Nursing
An ideal diploma program for anyone beginning their nursing career or wanting to continue their education in nursing.

Psychology Transfer Pathway
You'll be prepared to take on advanced studies in psychology with this seamless transfer pathway degree.

Radiologic Technology
This competitive degree program will prepare you for a rewarding career in a high-demand health care specialty.

Sonography-Echocardiography Technology
Realize a rewarding, lucrative career in a specialty healthcare field with this hands-on degree program.

Sport Management
Turn your passion for athletics into a professional purpose – score a rewarding career in the sports industry with this degree program.
Surgical Technology
Train for a rewarding and in-demand career as a surgical technologist, caring for patients during the surgical process, with this degree program.

Survey Technician
Our Survey Technician certificate provides hands-on training with state-of-the-art equipment used for a wide variety of civil engineering and public works projects.

Visual Arts
Painting, photography, printmaking, ceramics and other forms of visual art are at the heart of this foundational certificate program.
Get Started

Thriving at M State
From fun student organizations and athletics to helpful resources and services, there’s a community waiting for you at M State.
How to Apply
We’re here for you. Get step-by-step guidance on requirements for admission and with the application process.

Cost and Financial Aid
M State has one of the lowest tuition rates in Minnesota. With financial aid, grants, scholarships and sponsorships, the future you imagine is within reach.
